KIT (Karlsruher Institut für Technologie) Zoologisches Institut Abteilung für Zell- und Neurobiologie |
 |
 |
 |
| Homepage |
| Mitarbeiter |
| Lehre |
| Forschung |
| Ausgewählte Publikationen |
| Diplomarbeiten, Zulassungsarbeiten, Doktorarbeiten |
| Anfahrt, Lageplan |
| » Zur AG Franz |
| » Zur Fakultät |
| » Zum CFN |
| » Zur KSOP |
» Zum KIT 
|
| Axon Guidance in the Retinotectal and in the Olfactory System | |
|
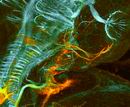
Zurück zur Übersicht "Forschung" Scientists involved: Martin Fritz, Dr. Christoph Gebhardt, Dr. Jana Ustinova, Markus Weschenfelder, Dr. Franco Weth, Prof. Dr. Martin Bastmeyer Funding: German Research Foundation (DFG), Karlsruhe School of Optics and Photonics (KSOP), Landesgraduiertenförderung (LGFG) Like in a computer's processor, all sensory, cognitive and motor capabilities of the brain rely on its connectional architecture, which is of bewildering complexity. In contrast to technical devices, this marvelous "computer" arises by self-organization during embryonic development. Outgrowing axons, forming the brain's wires, are thought to be guided to their appropriate targets by genetically encoded signals. The molecular mechanisms of these processes have remained poorly understood. Two very basic patterns of connectivity can be discerned - topographic and non-topographic connections. Topographic connections are used for the faithful transmission of preprocessed chunks of information from one brain center to the next. Non-topographic connections are used to re-sort information. As a model for the development of topographic connections we investigate the retinotectal projection transferring image information from the eye to the midbrain's optic tectum. Our model for non-topographic connections is the primary olfactory projection conveying odor-information from the nose to the forebrain's olfactory bulb.
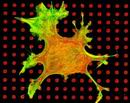
Scientists involved: Adrian Friebel, Dr. Christoph Gebhardt, Dr. Franco Weth, Prof. Dr. Martin Bastmeyer In this project, we analyze the development of the retinotectal projection through computational simulation in MATLAB and experimental validation. We have devised a theoretical model which implements all potentially relevant guidance cue interactions involved in this developmental process. The model is used to predict the outcomes of in-vitro assays of axonal guidance, which are subsequently experimentally realized. To this end, novel assay substrates of guidance molecules are fabricated using protein printing, microfluidics or photo-labile protein caging.  Project 2: Functional in vitro Analysis of Topographic Axon Guidance Scientists involved: Martin Fritz, Markus Weschenfelder, Dr. Franco Weth, Prof. Dr. Martin Bastmeyer  During topographic mapping, growth cones (the developing axon's steering device) perform precision navigation within fields
of external ephrin-A concentration gradients. Intriguingly, there is evidence that the same growth cones are able to desensitize
to this repulsive guidance factor, an observation that is hard to reconcile with topographic accuracy. We use micro-contact
printing and collapse assays to address the puzzle of growth cone adaptation and resensitization in response to ephrin-A5.
Using the so called "gap assay", we found that axons growing on homogeneous ephrin-A5 patterns become in fact desensitized,
but regain sensitivity after encountering an ephrin-A5-free stretch of way. To gather insights on the intracellular
mechanisms which are involved in this process, pharmacological inhibitors will be tested. In a second project, a method for
genetic manipulation of chick embryonic retinal explants by electroporation is established and optimized. With this method
we strive to introduce genetic sensors monitoring the dynamics of the molecular apparatus of axon guidance and to interfere
with its expression. Effects will be analyzed by in vitro experiments using defined patterns of guidance cues.
During topographic mapping, growth cones (the developing axon's steering device) perform precision navigation within fields
of external ephrin-A concentration gradients. Intriguingly, there is evidence that the same growth cones are able to desensitize
to this repulsive guidance factor, an observation that is hard to reconcile with topographic accuracy. We use micro-contact
printing and collapse assays to address the puzzle of growth cone adaptation and resensitization in response to ephrin-A5.
Using the so called "gap assay", we found that axons growing on homogeneous ephrin-A5 patterns become in fact desensitized,
but regain sensitivity after encountering an ephrin-A5-free stretch of way. To gather insights on the intracellular
mechanisms which are involved in this process, pharmacological inhibitors will be tested. In a second project, a method for
genetic manipulation of chick embryonic retinal explants by electroporation is established and optimized. With this method
we strive to introduce genetic sensors monitoring the dynamics of the molecular apparatus of axon guidance and to interfere
with its expression. Effects will be analyzed by in vitro experiments using defined patterns of guidance cues.
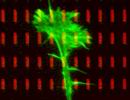
Project 3: Organization and Development of the Olfactory Projection in Fish and Mouse Scientists involved: Dr. Jana Ustinova, Dr. Franco Weth In the primary olfactory projection, in contrast to the retinotectal system, the spatial position of the parental neuron doesn't matter for the destination of its axon. Instead, axon targeting is determined by the specificity of the olfactory sensory neuron (OSN). Every sensory neuron expresses only one out of about 100 or 1000 olfactory receptor genes in zebrafish and mouse, respectively. OSNs of the same receptor phenotype are broadly scattered across the olfactory epithelium but all converge their axon to the same stereotypic point on the olfactory bulb. In the zebrafish, we investigate how, despite of a global randomness, subtle differences in the spatial distributions of OSN types contribute to axon sorting. In the mouse, we have embarked on a de novo search for genes involved in OSN axon targeting by differential screening of single-cell cDNA libraries of outgrowing OSNs of defined specificity. Collaboration partners: Dr. Jörg Strotmann (University of Hohenheim) and Dr. Natja Haag (former collaborator)  | |
|---|---|